The following plot shows the variation of first ionization energies across the periodic table. Consistent with size trends, first ionization energies generally increase across a period and decrease down a group. The valence electrons are closer to the nucleus to which they are attracted in a smaller atom thus, more energy will be required to remove an electron by ionization. Worksheets include both questions and answers. Trends in first ionization energies can be understood on the basis of size of atoms. This entry was posted on by Anne Helmenstine (updated on June 6, 2022) These element names and symbols worksheets help you practice identifying the one- and two-letter element symbols and the names of the elements of the periodic table. \Īll ionization energies are positive, because it takes energy to remove an electron from the attraction of an atom’s nucleus. Each represented one of the 63 elements known at the time. To put some order into his study of chemical elements, Mendeleev made up a set of cards. In the game, cards are arranged both by suit, horizontally, and by number, vertically. 
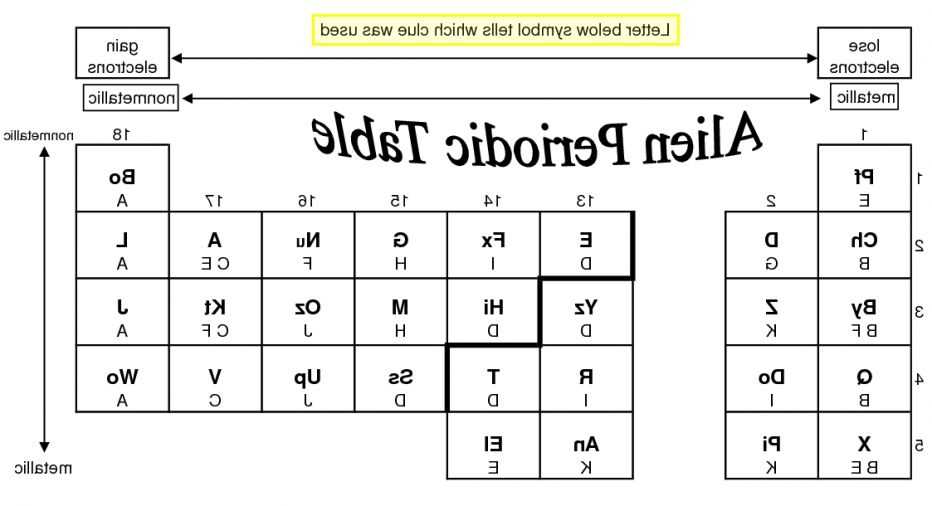
Vul cania (V) is in the same period as quackzil (Q) and wobble (Wo). Mendeleev was said to have been inspired by the card game Solitaire. The first ionization energy, \(I_1\), refers to removing one electron from a neutral atom: The halogens (group 17) are apstrom (A), vulcania (V), and kratt (K t). Ionization energy, I, is the energy required to remove an electron from a gaseous species. Our chemistry worksheets are designed to give students valuable information while also challenging them in areas that. You’ll easily find worksheets on a variety of chemistry topics, according to the NCERT syllabus. In the chart below, note the trends in the sizes (radii in picometers, pm) of the following ions with the same charge. Worksheet 5 : Types of elements and Properties Topic 2: The Periodic Table Set A: Terms and Definitions Objective: By defining these words, you should become more familiar with Periodic Table related terms and their definitions Define, neatly and clearly, the following Periodic Table related terms. The set of chemistry worksheets will help pupils understand the most crucial concepts they’ll need to succeed in their studies. 
Procedure Fill in the blank periodic table below with the correct alien planet symbol for each element. Pie loses them from its fifth energy level, while zapper loses them from its third. The elements zapper (Z) and pie (Pi) both lose 2 electrons. Ionic radii increase down a group for ions of the same charge. The element anatom (An) tends to lose 3 electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
